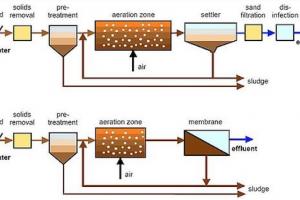
Find Dissolved Oxygen in given Sample by Azide Modification
Reactants:
-
MnSO4
-
Alkali
-
Iodide Azide (NaoH + NaH3 + NaI)
-
H2SO4 conc.
-
Starch Indicaoter,
-
Na2S203(N=0.025)
-
Oxygen is required for all living organisms for growth (metabolism) 21% in air quantity directly related with atm pressure and inversly proportional to temp for trout 7.5 mgl required
-
BOD (vol= 300 ml)
Procedure:
-
Add 2ml alkali iodide azide if becomes yellow = oxygen present while no oxygen ppt will be created let it settle ( Na2S03, Sodium sulphride) brings oxygen to zero
-
Add NaSO3 to another sample (oxygen become zero)
-
Add MnSO4 add alkali iodide axide color while means no oxygen.
-
Add 2ml H2SO4 ro disolve (in first sample) color becomes as mastard oil
-
Remove 100ml from the sample
-
Add 1ml starch indicator to the remaining sample => color = blueish
-
Take NaS2o3 in burrette
-
Titrate the sample against it until it becomes colorless ==> initial reading=4ml ==> final reading=12.6ml ==> 12.6-4=8.6ml
-
++ (oH) 1ml of Na2SO3 = 1mgk of dissolved oxygen it contains 8-6 mgk of dissolved oxygen Mn + H2o => M(oH)2
-
Mn (oH)2 + 1/2 o2 =>Mno2+H2o
-
Mno2 + 2i + 4H + => Mn + i2 +2H2o